We can't find the internet
Attempting to reconnect
Something went wrong!
Hang in there while we get back on track

Bennett Capital Partners
Bennett Capital Partners is your trusted mortgage broker in Miami, FL, offering home loans, refinancing, and investment property loans wi...
Miami, Florida

Portland Chauffeur
Experience premium private town car and chauffeur services in Portland. Offering reliable PDX airport transportation and executive black ...
Portland, Oregon

Achievable
Achievable bietet die effektivste Methode, um professionelle Prüfungen zu bestehen und Ihre Karriere voranzutreiben. Lernen Sie unterwegs...
Online

Rikayaa Enterprises Ltd.
Rikayaa Enterprises Ltd. is a premier manufacturer of tin ingots, lead, and tin-lead alloys, committed to sustainability and innovation. ...
Not specified
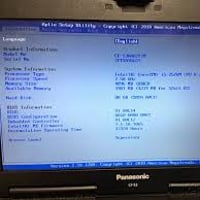
Rapid BIOS Password Recovery
Expert BIOS password recovery for all Panasonic Toughbook models. Fast, reliable service with guaranteed results. Unlock your Toughbook n...
United States
Thai Kru
Your Thailand Travel guru for vacations and relocation. Visas, Condos, Tours and More.
Thailand

